
- 收藏
- 加入书签
固体燃料热化学转化的反应分子动力学模拟进展
摘要:为进一步优化和改进煤炭和生物质等固体燃料的转化流程,提高能源利用效率,探究固体燃料的热化学转化机理成为当然能源领域研究的重点之一 。实验难以捕捉高温瞬时的自由基反应,而数值模拟方法可以获取反应过程中的细节。因此,反应分子动力学(ReaxFF molecular dynamics, ReaxFF MD)模拟被广泛地应用于研究煤等固体燃料的转化机理。本文介绍了ReaxFF 反应力场的建立和发展历程,揭示了其在重现复杂化学反应方面的优势。随后,讨论了ReaxFF MD 在煤热解和气化领域的应用,其中煤模型的规模、模拟温度和模拟时间会对模型的转化过程产生不同影响。此外,还探究了木质纤维素类生物质中利用ReaxFF MD 方法来揭示产物生成路径和气化机理等方面的有效性。
关键词:固体燃料;反应分子动力学;煤炭;热化学反应
中图分类号:O6 文献标识码:A
0 引言
当前,煤炭在我国能源结构中占有重要地位。随着我国对可持续发展与双碳目标的日益关注,玉米、小麦秸秆以及烟草梗等农业废弃物因其来源广泛与碳中和特性成为替代传统化石能源的重要选择[1]。固体燃料的热转化方法包括热解、气化和燃烧等类型。由于其热化学反应的环境极其复杂且涉及许多耦合反应途径,通过实验很难检测高温下瞬时的自由基反应,因此目前对固体燃料的热化学反应机理尚缺乏详细的认识。利用分子动力学的方法模拟固体燃料的热化学反应过程,分析其反应机理逐渐成为主流。反应分子动力学方法(ReaxFF MD)结合了量子化学和传统的非反应经验力场的分子动力学方法,旨在利用可变势能来描述含有键的形成和断裂的化学反应。在过去的二十多年反应分子动力学方法被广泛应用于揭示各类固体燃料的热化学反应机理。本文简述了反应分子动力学的发展历程,介绍了反应分子动力学在分析煤和生物质等类煤有机物热化学反应机理中的应用与潜力。
1 反应分子动力学(ReaxFF MD)的发展
反应分子动力学是一种针对大规模的分子体系,更高效地重现其化学反应的分子动力学方法。传统的经典分子动力学方法只能从分子尺度模拟体系的物理现象和过程,对于化学键断裂等现象无法描述。而量子化学方法可以模拟分子体系中化学键的断裂和形成、化学反应途径等过程,但计算代价相对更高,不适用于大规模体系的模拟[2]。
因此,van Duin  人[3]建立了基于键级的ReaxFF 反应力场来解决这一问题。在ReaxFF 反应力场中,键级与原子间的距离有关。随着原子间距的增大,原子的势能会平滑地趋于零,因而整个体系的能量是连续的。由原子间距离描述的键级表达如下[3]:
人[3]建立了基于键级的ReaxFF 反应力场来解决这一问题。在ReaxFF 反应力场中,键级与原子间的距离有关。随着原子间距的增大,原子的势能会平滑地趋于零,因而整个体系的能量是连续的。由原子间距离描述的键级表达如下[3]:
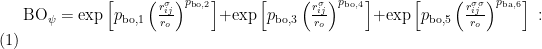
其中,BOij 表示原子i 和原子j 之间的键级,ro 是平衡键长,rij 表示原子间距离,pbo 表示相关的经验参数。此外,原子间的非键相互作用被归纳为库伦相互作用和范德华相互作用。整个系统的能量方程如下:
Esystem=Ebond+Eover+Eunder+Elp+Eval+Etor+EvdWals+ECoulomb(
其中,Ebond 表示键能;Eover 表示过配位原子的校正能;Eunder 表示欠配位原子的校正能;Eval 表示共价键角能;Epen 表示两个双键以键角共用一个原子产生的额外能量损失;Etors 表示二面角作用能;Econj 表示共轭作用能;EvdWaals 和ECoulomb 分别表示范德华相互作用和库伦相互作用。该力场被用来模拟烃类系统,证明了ReaxFF 力场在模拟复杂反应系统方面的优势。
随后,van Duin 等人[4]建立了 Si/H/O 的反应力场来预测硅及其氧化物材料的结构、性质和界面反应等,实现了精确的动态模拟效果。之后更多的研究人员参与到各类反应力场参数集的建立工作中,反应力场的参数集拓展到了N[5]、S[6]等非金属元素和 Al[7] 、Cu[8]、Li[9]、 ΔMg[10] 和Mo[11]等金属元素。迄今为止,已有超过五十种元素的相关反应力场被开发出来,其模拟应用的范围涉及碳氢化合物及煤的热解与气化[12–15]和新材料的开发[16]等多个方面。
2 ReaxFF 反应力场在煤的热化学反应中的应用
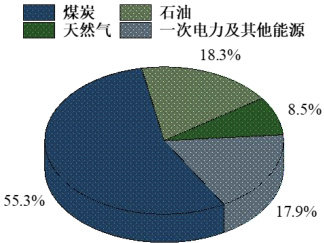
煤炭等化石能源在我国能源消费结构中占有举足轻重的地位。根据国家统计局数据[17],近十年来我国煤炭消费在能源消费总量中占比持续超过一半以上。如图1 所示,2023 年我国能源消费结构统计中,化石燃料的占比达到 82.1% ,其中煤炭占全年能源消耗的 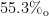 煤炭的利用技术主要包括热解、气化和燃烧等过程。通过热解处理,煤炭可转化为焦炭和焦油等化学原料,而气化技术则能将煤炭转化为合成气,并有效控制污染物排放,提供清洁和灵活的能源利用方式。
煤炭的利用技术主要包括热解、气化和燃烧等过程。通过热解处理,煤炭可转化为焦炭和焦油等化学原料,而气化技术则能将煤炭转化为合成气,并有效控制污染物排放,提供清洁和灵活的能源利用方式。
近年来,对煤炭热解和气化的研究主要集中在热解产物的分布、主要化学结构的演化和气化剂及矿物组分的影响机理等 18,19] 。尽管大量的实验从宏观视角提供了详细的观测结果,但对于反应机理以及复杂的分子间相互作用等微观细节的深入分析则显得困难。相比之下,反应分子动力学方法能够准确描述化学键的断裂、形成和重排等过程,从而揭示煤炭热转化过程中的反应路径、自由基以及中间产物的生成和消耗和结构演化等细节。
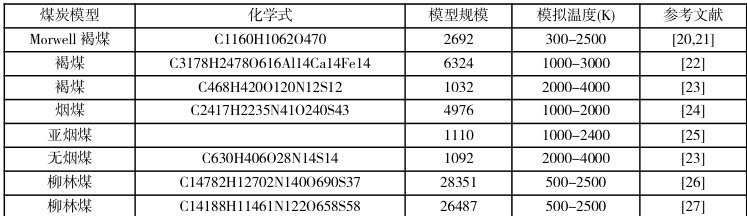
煤炭分子的结构对其热转化有着显著影响,特定结构的分子模型可以影响失重曲线变化和产物的种类及其分布。Salmon 等[20]构建了一个Morwell 褐煤大分子模型,探究了不同升温速率下褐煤模型的热解规律。研究证明,ReaxFF MD 能够再现各种实验研究中观察到的大分子的脱官能团、解聚和残基重排等过程。证明ReaxFFMD 可用于模拟探索复杂的凝聚相热解分子动力学模型。表 1 中列出了部分ReaxFF 模拟研究中采用和构建的煤分子模型及其模拟条件。可以看出煤模型规模集中在几千到几万原子级别,通常认为小规模的模型可以研究主要热解产物的生成路径和分布,但难以反应煤内部结构的复杂性。大规模分子模型可以探索煤热化学反应过程中主要化学键和官能团的断裂过程,为解释煤反应过程的机理提供支持。
表 1 不同类型煤的分子模型和应用条件

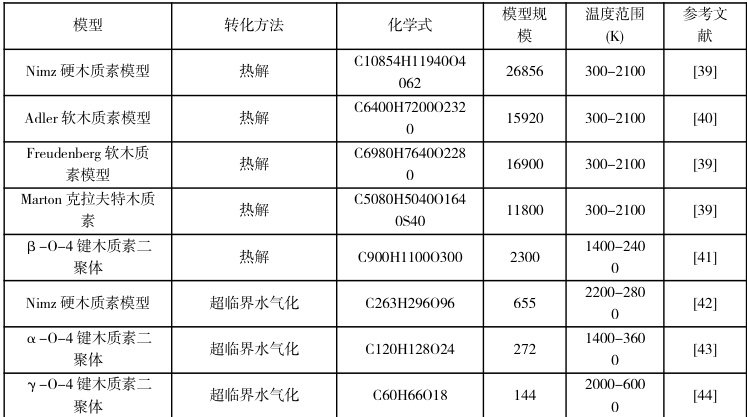
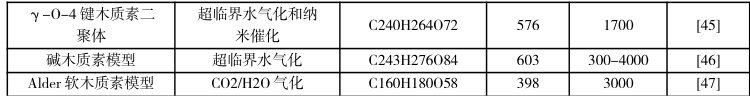
综上,ReaxFF在生物质领域的应用展现了其在复杂反应体系研究中的独特优势。通过深入揭示生物质及其主要组分热化学转化的反应机理,优化热化学过程,探索催化剂作用原理,ReaxFF为生物质的高效利用和清洁能源技术的发展提供了强有力的理论支持。
4 结论
ReaxFF MD方法在探究能源转化机理方面取得了显著成就。本文介绍了ReaxFF反应力场的建立和模拟原理,简述了反应力场的发展历程。经过二十多年的发展,当前已有五十多种元素的相关反应力场被开发出来,广泛应用在各类材料的开发和能源领域。其中,在揭示煤和生物质等固体燃料裂解、气化及燃烧过程中复杂的化学反应路径和中间产物的演化规律等方面取得了诸多研究成果。此外,ReaxFF MD方法可以为各类农业废弃物的热化学处理方面提供理论支持与指导。利用ReaxFF MD模拟农业废弃物热转化过程中的自由基反应,获取不同工艺条件下的产物分布,对设计高效清洁的能源转化技术,提高废弃物转化效率以及降低污染物排放具有重要意义。
参考文献
[1]C.C. Chang, R. Li, Agricultural waste, Water Environ. Res. 91 (2019) 1150–1167. https://doi.org/10.1002/wer.1211.
[2]R.A. Friesner, Ab initio quantum chemistry: Methodology and applications, Proc. Natl. Acad. Sci. U. S. A. 102 (2005) 6648–6653. https://doi.org/10.1073/pnas.0408036102.
[3]A.C.T. Van Duin, S. Dasgupta, F. Lorant, W.A. Goddard, ReaxFF: A reactive force field for hydrocarbons, J. Phys. Chem. A 105 (2001) 9396–9409. https://doi.org/10.1021/jp004368u.
[4]A.C.T. Van Duin, A. Strachan, S. Stewman, Q. Zhang, X. Xu, W.A. Goddard, ReaxFFSiO reactive force field for silicon and silicon oxide systems, J. Phys. Chem. A 107 (2003) 3803–3811. https://doi.org/10.1021/jp0276303.
[5]A. Strachan, A.C.T. van Duin, D. Chakraborty, S. Dasgupta, W.A. Goddard, Shock waves in high-energy materials: The initial chemical events in nitramine RDX, Phys. Rev. Lett. 91 (2003) 7–10. https://doi.org/10.1103/PhysRevLett.91.098301.
[6]T.T. Järvi, A.C.T. Van Duin, K. Nordlund, W.A. Goddard, Development of interatomic ReaxFF potentials for Au-S-C-H systems, J. Phys. Chem. A 115 (2011) 10315–10322. https://doi.org/10.1021/jp201496x.
[7]Q. Zhang, T. Çağln, A. Van Duin, W.A. Goddard, Y. Qi, L.G. Hector, Adhesion and nonwetting-wetting transition in the Al/α−Al2O3 interface, Phys. Rev. B - Condens. Matter Mater. Phys. 69 (2004) 1–11. https://doi.org/10.1103/PhysRevB.69.045423.
[8]K.D. Nielson, A.C.T. Van Duin, J. Oxgaard, W.Q. Deng, W.A. Goddard, Development of the ReaxFF reactive force field for describing transition metal catalyzed reactions, with application to the initial stages of the catalytic formation of carbon nanotubes, J. Phys. Chem. A 109 (2005) 493–499. https://doi.org/10.1021/jp046244d.
[9]S.S. Han, A.C.T. Van Duin, W.A. Goddard, H.M. Lee, Optimization and application of lithium parameters for the reactive force field, ReaxFF, J. Phys. Chem. A 109 (2005) 4575–4582. https://doi.org/10.1021/jp051450m.
[10]S. Cheung, W.Q. Deng, A.C.T. Van Duin, W.A. Goddard, ReaxFF MgH reactive force field for magnesium hydride systems, J. Phys. Chem. A 109 (2005) 851–859. https://doi.org/10.1021/jp0460184.
[11]W.A. Goddard, A. Van Duin, K. Chenoweth, M.J. Cheng, S. Pudar, J. Oxgaard, B. Merinov, Y.H. Jang, P. Persson, Development of the ReaxFF reactive force field for mechanistic studies of catalytic selective oxidation processes on BiMoOx, Top. Catal. 38 (2006) 93–103. https://doi.org/10.1007/s11244-006-0074-x.
[12]E. Salmon, A.C.T. van Duin, F. Lorant, P.-M. Marquaire, W.A. Goddard, Early maturation processes in coal. Part 2: Reactive dynamics simulations using the ReaxFF reactive force field on Morwell Brown coal structures, Org. Geochem. 40 (2009) 1195–1209. https://doi.org/https://doi.org/10.1016/j.orggeochem.2009.09.001.
[13]F. Castro-Marcano, A.M. Kamat, M.F. Russo, A.C.T. van Duin, J.P. Mathews, Combustion of an Illinois No. 6 coal char simulated using an atomistic char representation and the ReaxFF reactive force field, Combust. Flame 159 (2012) 1272–1285. https://doi.org/10.1016/j.combustflame.2011.10.022.
[14]F. Castro-Marcano, V. V. Lobodin, R.P. Rodgers, A.M. McKenna, A.G. Marshall, J.P. Mathews, A molecular model for Illinois No. 6 Argonne Premium coal: Moving toward capturing the continuum structure, Fuel 95 (2012) 35–49. https://doi.org/10.1016/j.fuel.2011.12.026.
[15]F. Castro-Marcano, M.F. Russo, A.C.T. Van Duin, J.P. Mathews, Pyrolysis of a large-scale molecular model for Illinois no. 6 coal using the ReaxFF reactive force field, J. Anal. Appl. Pyrolysis 109 (2014) 79–89. https://doi.org/10.1016/j.jaap.2014.07.011.
[16]E. Zaminpayma, K. Mirabbaszadeh, Interaction between single-walled carbon nanotubes and polymers: A molecular dynamics simulation study with reactive force field, Comput. Mater. Sci. 58 (2012) 7–11. https://doi.org/10.1016/j.commatsci.2012.01.023.
[17]中华人民共和国国家统计局, 中国统计年鉴, 2024 (n.d.). https://www.stats.gov.cn/sj/ndsj/2024/indexch.htm.
[18]C. Geng, S. Li, C. Yue, Y. Ma, Pyrolysis characteristics of bituminous coal, J. Energy Inst. 89 (2016) 725–730. https://doi.org/10.1016/j.joei.2015.04.004.
[19]P. Zhu, Z. Yu, J. Zhang, B. Dai, J. Zhang, P. Liang, Z. Lei, Catalytic Pyrolysis of Bituminous Coal under Pyrolysis Gas over a Ni/MgO Catalyst, Chem. Eng. Technol. 40 (2017) 1605–1610. https://doi.org/10.1002/ceat.201700163.
[20]E. Salmon, A.C.T. van Duin, F. Lorant, P.M. Marquaire, W.A. Goddard, Early maturation processes in coal. Part 2: Reactive dynamics simulations using the ReaxFF reactive force field on Morwell Brown coal structures, Org. Geochem. 40 (2009) 1195–1209. https://doi.org/10.1016/j.orggeochem.2009.09.001.
[21]G. Yan, Z. Zhang, K. Yan, Reactive molecular dynamics simulations of the initial stage of brown coal oxidation at high temperatures, Mol. Phys. 111 (2013) 147–156. https://doi.org/10.1080/00268976.2012.708443.
[22]G.Y. Li, J.X. Ding, H. Zhang, C.X. Hou, F. Wang, Y.Y. Li, Y.H. Liang, ReaxFF simulations of hydrothermal treatment of lignite and its impact on chemical structures, Fuel 154 (2015) 243–251. https://doi.org/10.1016/j.fuel.2015.03.082.
[23]S. Bhoi, T. Banerjee, K. Mohanty, Insights on the combustion and pyrolysis behavior of three different ranks of coals using reactive molecular dynamics simulation, RSC Adv. 6 (2016) 2559–2570. https://doi.org/10.1039/c5ra23181g.
[24]M. Zheng, X. Li, J. Liu, L. Guo, Initial chemical reaction simulation of coal pyrolysis via ReaxFF molecular dynamics, Energy Fuels 27 (2013) 2942–2951. https://doi.org/10.1021/ef400143z.
[25]J.H. Zhan, R. Wu, X. Liu, S. Gao, G. Xu, Preliminary understanding of initial reaction process for subbituminous coal pyrolysis with molecular dynamics simulation, Fuel 134 (2014) 283–292. https://doi.org/10.1016/j.fuel.2014.06.005.
[26]M. Zheng, X. Li, F. Nie, L. Guo, Investigation of Overall Pyrolysis Stages for Liulin Bituminous Coal by Large-Scale ReaxFF Molecular Dynamics, Energy Fuels 31 (2017) 3675–3683. https://doi.org/10.1021/acs.energyfuels.6b03243.
[27]M. Zheng, X. Li, L. Guo, Investigation of N behavior during coal pyrolysis and oxidation using ReaxFF molecular dynamics, Fuel 233 (2018) 867–876. https://doi.org/10.1016/j.fuel.2018.06.133.
[28]D. Hong, X. Guo, Molecular dynamics simulations of Zhundong coal pyrolysis using reactive force field, Fuel 210 (2017) 58–66. https://doi.org/10.1016/j.fuel.2017.08.061.
[29]M. Gao, X. Li, L. Guo, Pyrolysis simulations of Fugu coal by large-scale ReaxFF molecular dynamics, Fuel Process. Technol. 178 (2018) 197–205. https://doi.org/10.1016/j.fuproc.2018.05.011.
[30]M. Zheng, X. Li, M. Wang, L. Guo, Dynamic profiles of tar products during Naomaohu coal pyrolysis revealed by large-scale reactive molecular dynamic simulation, Fuel 253 (2019) 910–920. https://doi.org/10.1016/j.fuel.2019.05.085.
[31]F. Xu, H. Liu, Q. Wang, S. Pan, D. Zhao, Q. Liu, Y. Liu, ReaxFF-based molecular dynamics simulation of the initial pyrolysis mechanism of lignite, Fuel Process. Technol. 195 (2019) 106147. https://doi.org/10.1016/j.fuproc.2019.106147.
[32]中国产业发展促进会, 2024中国生物质能产业发展年度报告, (2024). https://beipa.org.cn/newsinfo/7147401.html.
[33]H.A.N. Lu, W. Chao, Z. Wentao, W. Xu, D.O.U. Yuqing, Y. Jing, T.A.N. Jianeng, Effect of heating pretreatment on pyrolysis and sensory quality of heated cigarette flakes, Chinese Tob. Sci. 46 (2025) 60–68. https://doi.org/10.13496/j.issn.1007-5119.2025.02.008.
[34]Y. Dai, J. Xu, L. Zhu, J. Jiang, Y. Zhou, G. Zhou, Mechanism study on the effect of glycerol addition on tobacco pyrolysis, J. Anal. Appl. Pyrolysis 157 (2021) 105183. https://doi.org/10.1016/j.jaap.2021.105183.
[35]P. McKendry, Energy production from biomass (part 1): Overview of biomass, Bioresour. Technol. 83 (2002) 37–46. https://doi.org/10.1016/S0960-8524(01)00118-3.
[36]M. Zheng, Z. Wang, X. Li, X. Qiao, W. Song, L. Guo, Initial reaction mechanisms of cellulose pyrolysis revealed by ReaxFF molecular dynamics, Fuel 177 (2016) 130–141. https://doi.org/10.1016/j.fuel.2016.03.008.
[37]Z. Liu, X. Ku, H. Jin, S. Yang, Research on the microscopic reaction mechanism of cellulose pyrolysis using the molecular dynamics simulation, J. Anal. Appl. Pyrolysis 159 (2021) 105333. https://doi.org/10.1016/j.jaap.2021.105333.
[38]Y. Feng, H. Hao, C.L. Chow, D. Lau, Exploring reaction mechanisms and kinetics of cellulose combustion via ReaxFF molecular dynamics simulations, Chem. Eng. J. 488 (2024). https://doi.org/10.1016/j.cej.2024.151023.
[39]T. Zhang, X. Li, L. Guo, X. Guo, Reaction mechanisms in pyrolysis of hardwood, softwood, and kraft lignin revealed by ReaxFF MD simulations, Energy Fuels 33 (2019) 11210–11225. https://doi.org/10.1021/acs.energyfuels.9b02843.
[40]T. Zhang, X. Li, X. Qiao, M. Zheng, L. Guo, W. Song, W. Lin, Initial mechanisms for an overall behavior of lignin pyrolysis through large-scale ReaxFF molecular dynamics simulations, Energy Fuels 30 (2016) 3140–3150. https://doi.org/10.1021/acs.energyfuels.6b00247.
[41]Y. Zhou, Q. Dang, Y. Wu, T. Lei, A mechanistic investigation of lignin dimer fast pyrolysis from reactive molecular dynamics simulation, J. Environ. Chem. Eng. 9 (2021). https://doi.org/10.1016/j.jece.2021.106484.
[42]H. Li, B. Xu, H. Jin, K. Luo, J. Fan, Molecular dynamics investigation on the lignin gasification in supercritical water, Fuel Process. Technol. 192 (2019) 203–209. https://doi.org/10.1016/j.fuproc.2019.04.014.
[43]Z. Song, M. Bai, Z. Yang, H. Lei, M. Qian, Y. Zhao, R. Zou, C. Wang, E. Huo, Gasification of α-O-4 linkage lignin dimer in supercritical water into hydrogen and carbon monoxide: Reactive molecular dynamic simulation study, Fuel 329 (2022) 125387. https://doi.org/10.1016/j.fuel.2022.125387.
[44]X. Liu, T. Wang, J. Chu, M. He, Q. Li, Y. Zhang, Understanding lignin gasification in supercritical water using reactive molecular dynamics simulations, Renew. Energy 161 (2020) 858–866. https://doi.org/10.1016/j.renene.2020.06.022.
[45]T. Wang, X. Liu, H. Liu, M. He, Synergistic effect of supercritical water and nano-catalyst on lignin gasification, Int. J. Hydrogen Energy 46 (2021) 34626–34637. https://doi.org/10.1016/j.ijhydene.2021.08.061.
[46]J. Chen, C. Wang, W. Shang, Y. Bai, X. Wu, Study on the mechanisms of hydrogen production from alkali lignin gasification in supercritical water by ReaxFF molecular dynamics simulation, Energy 278 (2023) 127900. https://doi.org/10.1016/j.energy.2023.127900.
[47]Y. Pang, X. Zhu, N. Li, Z. Wang, Investigation on reaction mechanism for CO2 gasification of softwood lignin by ReaxFF MD method, Energy 267 (2023) 126533. https://doi.org/10.1016/j.energy.2022.126533.
作者简介:吴育洁(1993—),女,助理工程师,硕士研究生,主要研究方向为材料化学。
通信作者:于永龙(1981—),男,大学本科,主要研究方向为生产制造。





 京公网安备 11011302003690号
京公网安备 11011302003690号


